Real patients, real responses: Timed 25-Foot Walk (T25FW) on video
Before and after: see how these patients responded to AMPYRA® (dalfampridine) as they performed the Timed 25-Foot Walk (T25FW) test. In the clinical studies, the efficacy of AMPYRA was shown by an increase in walking speed, as measured by the T25FW using a responder analysis (See Results and Description of Studies in Efficacy in 2 clinical studies). Walking speed has been demonstrated as a useful and reliable functional measure of walking ability.10
Selected Important Safety Information
Concomitant use with OCT2 inhibitors (e.g., cimetidine) may cause increased exposure to AMPYRA and potential risk of seizures.
For additional Important Safety Information, please see below.
Please ensure that your volume is turned up when viewing the video.
Not every patient responds to AMPYRA. Individual patient response to therapy may vary.
View More Patient Videos
-
 Susan
SusanMS Type: Relapsing remitting Age: 60 years Gender: Female Occupation: Volunteer MS Disease Duration: Not reported Time on Treatment: 9 weeks Baseline Walking Time: 10.74 sec Baseline Walking Speed: 2.33 fps Walking Time After 9 Weeks on Treatment: 8.28 sec Walking Speed After 9 Weeks on Treatment: 3.02 fps Average Increase in Speed: 29.61% 
-
 Nick
NickMS Type: Secondary progressive Age: 58 years Gender: Male Occupation: Lumber Sales MS Disease Duration: 11 years Time on Treatment: 6 weeks Baseline Walking Time: 10.22 sec Baseline Walking Speed: 2.45 fps Walking Time After 6 Weeks on Treatment: 7.86 sec Walking Speed After 6 Weeks on Treatment: 3.18 fps Average Increase in Speed: 29.80% 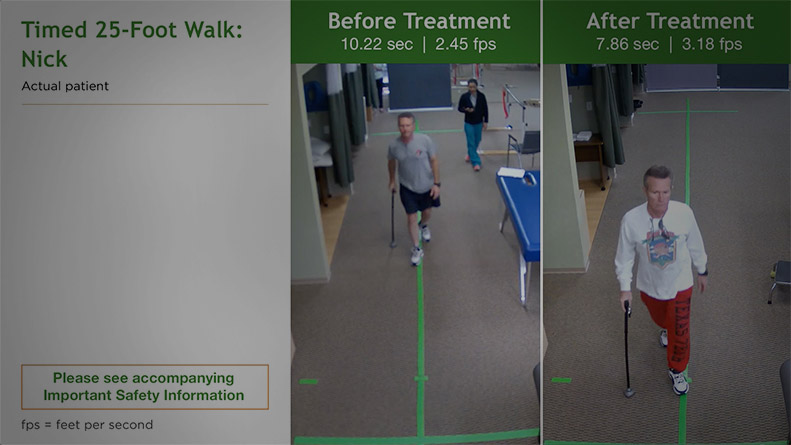
-
 Lisa
LisaMS Type: Relapsing remitting Age: 42 years Gender: Female Occupation: N/A MS Disease Duration: 14 years Time on Treatment: 12 weeks Baseline Walking Time: 11.26 sec Baseline Walking Speed: 2.22 fps Walking Time After 12 Weeks on Treatment: 8.80 sec Walking Speed After 12 Weeks on Treatment: 2.84 fps Average Increase in Speed: 27.93% 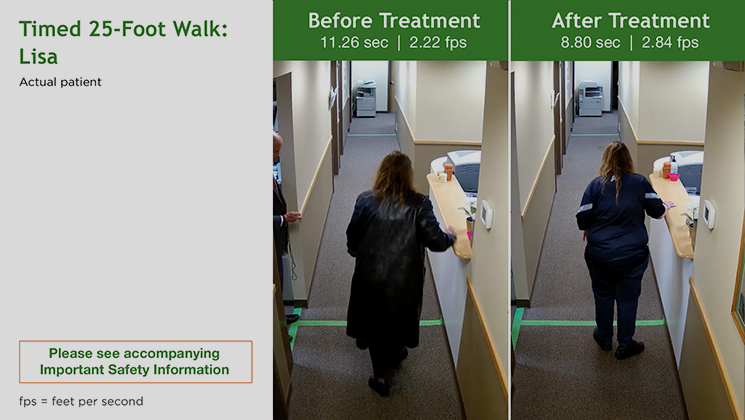
Indication
AMPYRA® (dalfampridine) Extended Release Tablets, 10 mg, is indicated to improve walking in adults with multiple sclerosis (MS). This was demonstrated by an increase in walking speed.
Important Safety Information
AMPYRA is contraindicated in patients with history of seizure, moderate or severe renal impairment (CrCl ≤ 50 mL/min), or history of hypersensitivity to AMPYRA or 4-aminopyridine.
Indication
AMPYRA® (dalfampridine) Extended Release Tablets, 10 mg, is indicated to improve walking in adults with multiple sclerosis (MS). This was demonstrated by an increase in walking speed.
Important Safety Information
- AMPYRA is contraindicated in patients with history of seizure, moderate or severe renal impairment (CrCl ≤ 50 mL/min), or history of hypersensitivity to AMPYRA or 4-aminopyridine.
- AMPYRA can cause seizures. The risk of seizures increases with increasing doses. Permanently discontinue AMPYRA if seizure occurs. In the post-marketing period seizures have been reported. The majority of seizures occurred at the recommended dose, in patients without a history of seizures, and generally within days to weeks of starting therapy.
- AMPYRA has not been evaluated in patients with history of seizures or with epileptiform activity on an EEG, as these patients were excluded from clinical trials. The risk of seizures in patients with epileptiform activity on an EEG is unknown, and could be substantially higher than that observed in clinical studies.
- Avoid concomitant use of AMPYRA with other forms of 4-aminopyridine (4-AP, fampridine), since the active ingredient is the same. Instruct patients to discontinue use of any product containing 4-AP prior to initiating AMPYRA to reduce the potential for dose-related adverse reactions.
- AMPYRA can cause anaphylaxis and severe allergic reaction. Signs and symptoms included respiratory compromise, urticaria, and angioedema of the throat or tongue. If an anaphylactic or other serious allergic reaction occurs, permanently discontinue AMPYRA.
- AMPYRA is cleared predominantly by the kidneys. The risk of seizures in patients with mild renal impairment (CrCl 51–80 mL/min) is unknown, but AMPYRA plasma levels in these patients may approach those seen at a dose of 15 mg twice daily, a dose that may be associated with an increased risk of seizures. Estimated CrCl should be known before initiating AMPYRA and monitored at least annually during treatment.
- The most common adverse reactions (incidence ≥ 2% and at a rate greater than placebo) for AMPYRA in MS patients were urinary tract infection, insomnia, dizziness, headache, nausea, asthenia, back pain, balance disorder, MS relapse, paresthesia, nasopharyngitis, constipation, dyspepsia, and pharyngolaryngeal pain.
- The risk of adverse reactions, including seizures, increases with increasing AMPYRA doses. There is no evidence of additional benefit at doses greater than 10 mg twice daily.
- Concomitant use with OCT2 inhibitors (e.g., cimetidine) may cause increased exposure to AMPYRA and potential risk of seizures.
- There are no adequate data on AMPYRA in pregnant women. Based on animal data, use of AMPYRA during pregnancy may cause fetal harm.
- There are no data on presence of AMPYRA in breastmilk; benefits of breastfeeding should be considered along with benefit of AMPYRA to the mother and potential risks to the infant.
- Safety and effectiveness of AMPYRA in patients younger than 18 years have not been established.
- Clinical studies of AMPYRA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Because elderly patients are more likely to have decreased renal function, it is important to know the estimated CrCl before initiating AMPYRA.
Please see the Full Prescribing Information.
